The energy transition in key themes – a five part occasional series.
This will be short essays of five minutes, a thousand or so words and a chart meant to create a backdrop to all the other work I write here – origin stories as well as punditry if you will.
But beware – here there be equations.
1 – The Rest is Entropy
The Second Law of Thermodynamics
Let’s start at the very beginning, the death of the Universe and all that there is.
There are three laws of thermodynamics, which govern the creation and fate of energy in the universe (our universe at least – what a counter-factual to play with).
These are
The First Law – energy cannot be created, only changed from one form to another – (what we have we have)
The Second Law – you cannot get more energy from a system than you put in – at best you break-even (there are always losses) Except at absolute zero, the coldest temperature possible in the universe
The Third Law – you can’t reach absolute zero
There you have it.
Backing up, we have finite energy in the universe, we can transform but not add to it, and as we transform it we lose slivers of it so we tend toward zero energy over vast galaxies of time, slice by slice.
The big insight is the spoiler of the Second Law: although energy is all around us in various forms (heat of course, hence thermodynamics, but also electrical, mechanical, chemical and so on) –the big picture is since time began energy has been declining, always losing its original immensity, shape and sharpness tick by tick.
A burning candle is a beautiful night-light, but all that manufactured wax wanes as it dissipates fleeting heat then melts to a solid cold core.
And once gone, that energy cannot ever be fully rebuilt: the candle unable to be re-created from the ash.
Some great scientists studied such phenomenon and over decades mainly in the 19th century put philosophic narratives, then more precise numbers to it.
To them, The Second Law of Thermodynamics was developed in concept as an unending loss of energy as we used it, an unpayable energy debt we created as we consumed.
Place the First Law on top of it – we cannot create new energy, and the Third Law as a hard floor – we can only stop losing energy when we are infinitely dark and cold - –the Second Law tells us simply: what energy the Universe bequeathed us is all we have, and it is always running out.
Best, therefore, we use our time as energetic entities whether stars, planets or knowing humans wisely.
The scientists gave this insight a Greek word (and symbol S) and some key equations.
The word was entropy, from the Greek (of course) meaning transformation, and used it to link the ideas above to disorder, the loss of structure, the gradual deficiency of energy as it gets used, via friction, heat loss or emissions and other waste products.
And the equations were simple:
dS >= 0
meaning the change in energy disorder of a system –the Universe or a car engine - can only increase, never decrease. We always lose some energy when we use it, the only question is how much ?
How much debt are we drawing down each time we go to the vast but finite energy bank? We’ll put a number on that for our energy system today below.
But Ludwig Boltzmann in the 1890s put it another way, via probabilities or in its Sunday finery, Statistical Thermodynamics, which sought to answer fundamental questions such as what is temperature, how does heat energy get created for work – but also get wasted.
His tight mathematical summation:
S = k Log W
This reimagined disorder as inevitable as there are far far more ways for any system such as a gas or a physical being to be unstructured than existing as a single orderly unit.
Numerically, think playing cards thrown into the air landing randomly almost never falling in numbered order, milk poured into coffee unable to be pure milk again, the order of words in this essay able to be written in many billion ways, but you will only ever see this one version, the rest out there in a near-infinite jumbled heap.
This disorder equation is so profound, seeing the natural randomness of things, that it is inscribed on Boltzmann’s tombstone.
And so to our current energy system.
How do we fare against the gods of thermodynamics.
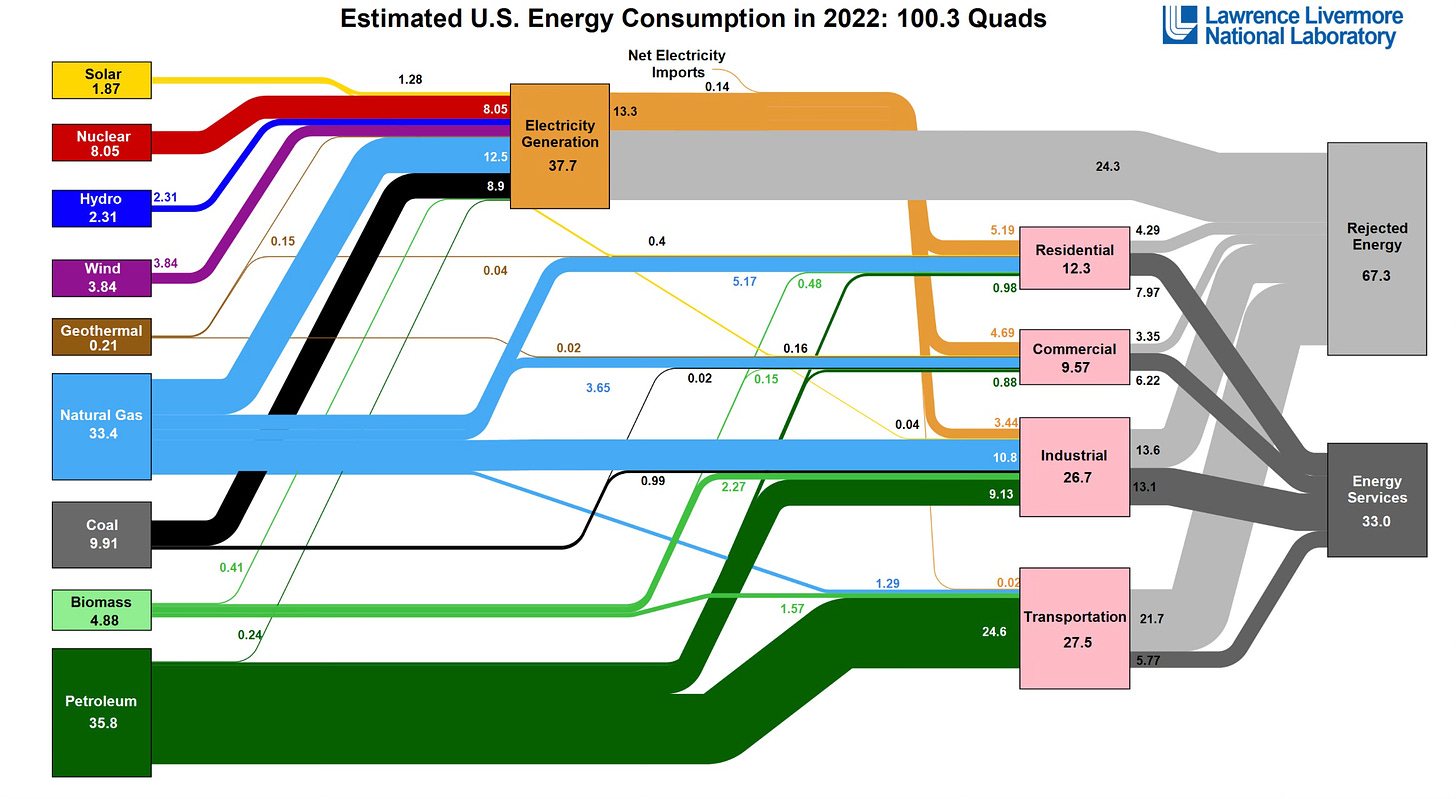
Let’s take a look at the classic way to assess our global energy use in a Sankey diagram (called after the lusciously named Irishman Matthew Henry Phineas Riall Sankey, who created it to look at the efficiency of steam engines).
As a simple way into this we can observe how the US used its energy in 2022 (as the US uses about one sixth of world energy this is a good approximation for the planet as a whole).
One does not need to be a student of Sankey diagrams or physical chemistry to understand the big picture here.
And note the unit of a “quad” or exajoule is the universal energy standard: each one is worth about 278 TWh (278 billion kWh) of electricity, or 178 million barrels of oil. Neatly, the US uses 100 of these units.
On the left side are all the main energy sources we have to hand: fossil fuels, nuclear, hydro-power, solar, wind and crucially electricity.
On the far right, the amount that is useful energy services such as heating, lighting and transport.
Take a moment to look at the far right of the picture.
33% of the energy created and supplied is ultimately used in final energy activity: 67% is “rejected energy” or lost energy.
In terms of the Second Law, we are wasting two-thirds of our energy via the thermal / fossil fuel system we currently use.
Feel free to roam the diagram, and look at each energy sector such as power and transport, but the big Sankey picture here is that two-thirds of the energy we “consume” today is unused.
We lose that two-thirds as waste heat and carbon dioxide mainly (because we use fossil fuels a lot).
In terms of the US although it consumes 100 quads of energy, it actually only requires 33 quads – the rest is entropy: waste, inefficiency, allowing the second law of thermodynamics to continue to win in a big way.
As long as we focus on fossil fuels as our burning energy source, this will be our energy fate.
Add to that, fossil fuels are finite, current reserves will last us about 50 years – less than a new lifetime, and heavily concentrated in just a few (Middle East) countries.
The reserves to production factor in Europe and the US for example is only about 10 years.
The gods of Second Law of Thermodynamics would at best give this energy system a C- grading: losses of over 66% !
But there is progress: in the Sankey diagram top left we have wind, solar and electricity: these are far more efficient energy systems, typically operating at 80% + efficiency, and now becoming energy mainstream: 15% global power, 6% primary energy.
And their reserves - sunlight and wind movement - are essentially infinite.
We’ll talk a lot more about these in the next few essays: how we are entering a new phase of energy development that will impress the Second Law gods in a much better way.
To end, when you hear talk of the energy transition therefore, a small plea that you don’t look upon it as political or local, but more universal, and governed by the Second Law.
We only need one-third of the energy we actually produce due to the second law of thermodynamics, and our over-devoted love of burning fossil fuels.
But we’ll fix that – more on this in the essays to come.